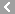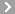CAS No.:64-47-1
Name: Physostigmine sulfate
Details Introduction
TOXICITY DATA with REFERENCE:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| dog | LDLo | subcutaneous | 1250ug/kg (1.25mg/kg) | LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Journal of Pharmacology and Experimental Therapeutics. Vol. 44, Pg. 95, 1932. |
| frog | LDLo | parenteral | 325mg/kg (325mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 44, Pg. 95, 1932. | |
| frog | LDLo | subcutaneous | 1gm/kg (1000mg/kg) | Naunyn-Schmiedeberg's Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 207, Pg. 547, 1949. | |
| man | TDLo | subcutaneous | 191ng/kg (0.000191mg/kg) | BEHAVIORAL: "HALLUCINATIONS, DISTORTED PERCEPTIONS" GASTROINTESTINAL: NAUSEA OR VOMITING GASTROINTESTINAL: OTHER CHANGES |
Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 53, Pg. 313, 1905. |
| mouse | LD50 | intraperitoneal | 510ug/kg (0.51mg/kg) | Archiv fuer Toxikologie. Vol. 29, Pg. 39, 1972. | |
| mouse | LD50 | intravenous | 178ug/kg (0.178mg/kg) | U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#03167, | |
| mouse | LD50 | oral | 1680ug/kg (1.68mg/kg) | "Psychotropic Drugs and Related Compounds," 2nd ed., Usdin, E., and D.H. Efron, Washington, DC, 1972Vol. -, Pg. 137, 1972. | |
| mouse | LD50 | subcutaneous | 700ug/kg (0.7mg/kg) | Annales Pharmaceutiques Francaises. Vol. 11, Pg. 286, 1953. | |
| rabbit | LDLo | subcutaneous | 1500ug/kg (1.5mg/kg) | LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Journal of Pharmacology and Experimental Therapeutics. Vol. 44, Pg. 95, 1932. |
| rat | LD50 | intraperitoneal | 1621ug/kg (1.621mg/kg) | Fundamental and Applied Toxicology. Vol. 4, Pg. S195, 1984. | |
| rat | LD50 | intravenous | 450ug/kg (0.45mg/kg) | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 259, Pg. 153, 1982. | |
| rat | LD50 | oral | 4mg/kg (4mg/kg) | Annales Pharmaceutiques Francaises. Vol. 11, Pg. 286, 1953. |
Consensus Reports:
EPA Genetic Toxicology Program.
SAFETY PROFILE:
Poison by subcutaneous, intravenous, and intraperitoneal routes. Moderately toxic by ingestion. Human systemic effects by subcutaneous route: hallucinations or distorted perceptions; nausea or vomiting, and other gastrointestinal changes. An experimental teratogen. Experimental reproductive effects. When heated to decomposition it emits toxic fumes of NOx and SOx. See also PHYSOSTIGMINE.
Hazard Codes of Physostigmine sulfate (CAS NO.64-47-1):  T+
T+
Risk Statements: 26/28
R26/28:Very toxic by inhalation and if swallowed.
Safety Statements: 25-45
S25:Avoid contact with eyes.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
Standards and Recommendations:
Analytical Methods:
Related Searches
Other Product
- -
- -
- -
- -
- -
- -
- -
- -
- -
- (4S,6R)-6-{2-[(2-hydroxy-ethyl)-(4-methoxy-benzenesulfonyl)-amino]-ethoxy}-4-isopropyl-5,6-dihydro-4H-pyran-2-carboxylic acid allyl ester
- [1,4'-Bipiperidine]-1'-carboxylic acid, 4-(1H-indol-7-yl)-, ethyl ester
- 1,3,5-Tris(4-acetamidophenyl)penta-1,5-dione
- 1,5-Bis(3-aminophenyl)-3-(4-N,N-di-methylaminophenyl)penta-1,5-dione
- 1,5-Bis(4-acetamidophenyl)-3-phenylpenta-1,5-dione
- 1,5-Bis(4-aminophenyl)-3-(4-acetamido-phenyl)penta-1,5-dione
- 1,5-Bis(4-aminophenyl)-3-phenylpenta-1,5-dione